Group of compounds containing hydrogen and carbon are known as hydrocarbons. They may be straight-chain, branched or cyclic compounds. Hydrocarbons naturally occur in coal, natural gas and many polymers like polystyrene, polythene, etc. When a compound has all the C-C single bonds it is known as saturated hydrocarbon (alkanes) and when double or triple bonds are present between C-C, it is known as an unsaturated hydrocarbon (alkene, alkyne). Various other functional groups (halide, hydroxyl, aldehyde, carboxylic, etc.) may be attached to the carbon in hydrocarbons.
1. Find the correct order of increasing acidity
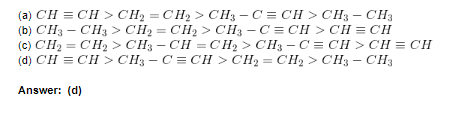
2. Identify Z in the given reaction:
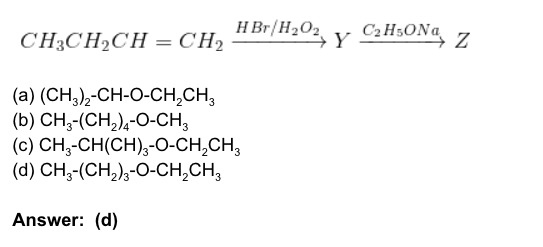
3. The product obtained on heating 2-Bromopentane with potassium ethoxide in ethanol is
(a) 1-pentene
(b) 2-cis-pentene
(c) trans-2-pentene
(d) 2-ethoxy pentane
Answer: (c)
4. Choose the process by which liquid hydrocarbons can be converted to gaseous hydrocarbons
(a) hydrolysis
(b) oxidation
(c) cracking
(d) distillation under reduced pressure
Answer: (c)
5. Which one of the following halide can be used in the Friedel-Crafts reaction?
(a) Isopropyl chloride
(b) Bromobenzene
(c) Chlorobenzene
(d) Chloroethene
Answer: (a)
6. Which one of the following compound forms salt on reaction with NaNH2?
(a) C2H2
(b) C2H6
(c) C6H6
(d) C2H4
Answer: (a)
7. Which among the following reagent can be used to distinguish between CH2BrCH=CH2 from CH3CH=CHBr?
(a) Br2/CCl4
(b) cold KMnO4
(c) AgNO3/C2H5OH
(d) Ag(NH3)2OH
Answer: (c)
8. Isopropyl iodide is formed after reaction of propene with HI, this is due to
(a) more stable free radical
(b) homolysis
(c) more stable carbanion
(d) more stable carbocation
Answer: (d)
9. C6H5CHO is formed when C6H6 is treated with CO and HCl in the presence of anhydrous AlCl3. Name of the reaction is:
(a) Friedel crafts reaction
(b) Gattermann Koch reaction
(c) Rosenmund reaction
(d) Stephen reaction
Answer: (b)
10. A compound having a bond angle 180° is
(a) alkyne
(b) alkane
(c) alkene
(d) cycloalkane
Answer: (a)
Recommended Video:

Comments