The three basic states in which a matter can exist are solid, liquid and gas. A matter can exist in one or more than one state, e.g. water exists in all three states. The state of a matter depends on the intermolecular forces and arrangement of molecules. The chemical property of a substance remains the same, irrespective of its state but the rate of reaction changes with the change in the physical state. On increasing and decreasing the temperature or pressure, one state of a matter gets converted to another state. Apart from these, there are two other states of matter known as Plasma and Bose-Einstein Condensate.
1. A container with a pin-hole contains equal moles of H2(g) and O2(g). Find the fraction of oxygen gas escaped at the same time when one-fourth of hydrogen gas escapes
(a) 1/16
(b) 1/4
(c) 1/2
(d) 1/8
Answer: (a)
2. What are the conditions for gas like Carbon monoxide to obey the ideal gas laws?
(a) low temperature and low pressure
(b) low temperature and high pressure
(c) high temperature and low pressure
(d) high temperature and high pressure
Answer: (c)
3. If the temperature is doubled, the average velocity of a gaseous molecule increases by
(a) 4
(b) 1.4
(c) 2
(d) 2.8
Answer: (b)
4. Find the molecular mass of a gas that takes three times more time to effuse as compared to He with the same volume
(a) 9 u
(b) 64 u
(c) 27 u
(d) 36 u
Answer: (d)
5. At the same temperature, the average molar kinetic energy of N2 and CO is
(a) KE1 > KE2
(b) KE1 < KE2
(c) KE1 = KE2
(d) insufficient information given
Answer: (c)
6. Find the temperature at which the rate of effusion of N2 is 1.625 times to that of SO2 at 500℃
(a) 620℃
(b) 173℃
(c) 110℃
(d) 373℃
Answer: (a)
7. Find the change in the root mean square speed of the gas on raising the temperature from 27℃ to 927℃
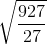
(a) becomes times
(b) gets doubled
(c) gets halved
(d) remains same
Answer: (b)
8. Find the relation between probable velocity, mean velocity and root mean square velocity
(a) 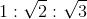
(b) 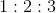
(c) 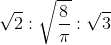
(d)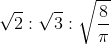
Answer: (c)
9. If 1.204 x 1021 molecules of H2SO4 are removed from 392 mg of H2SO4, find the moles of H2SO4 left.
(a) 4 x 10-3
(b) 1.5 x 10-3
(c) 1.2 x 10-3
(d) 2 x 10-3
Answer: (d)
10. Find the fraction of the total pressure exerted by hydrogen if it is mixed with ethane in an empty container at 25℃
(a) 15/16
(b) 1/16
(c) 1/2
(d) 1
Answer: (a)
Recommended Video:
Solid State Class 12 Chemistry One Shot & Mind Maps | NEET 2023 Exam Preparation

Comments