Alkyl halides are a part of the Organic Chemistry. Facts and reactions related to alkyl halides are asked every year in JEE. Some frequently asked questions related to alkyl halides are given here which may help you to prepare more effectively for JEE.
Frequently Asked Questions For Alkyl Halides
Question 1: What are alkyl halides?
Answer: Alkyl halides are organic compounds containing halogen atoms as a substituent. They are also known as haloalkanes, that is the compounds in which one or more hydrogen atoms in an alkane have been replaced by halogen atoms such as fluorine, chlorine, bromine or iodine.
Read More: Alkyl Halide
Question 2: Mention some of the physical properties of alkyl halides.
Answer: Some of the physical properties of alkyl halides are:
They have higher boiling point due to their high molecular weight. For a given alkyl group, the boiling point increases with increase in the atomic weight of the halogen, such that fluoroalkanes have the lowest boiling point and iodoalkanes have the highest boiling point. They are insoluble in water mainly due to their inability to form hydrogen bonds. They are soluble in typical organic solvents.
Question 3: What are the chemical properties of alkyl halides?
The lower members of alkyl halides are colourless, oily liquids with a sweet smell. Higher members of the group are solids.
They mainly undergo two types of reactions:
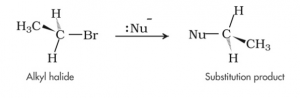
1. Nucleophilic Substitution reaction – Nucleophilic substitution of haloalkanes refers to the reaction in which halogens present in the haloalkanes are replaced by any other nucleophile.
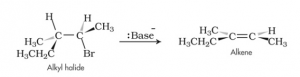
2.Elimination Reaction – Elimination reaction refers to a reaction in which two atoms are eliminated from a compound and an additional bond is formed. Since haloalkanes form highly polar covalent bonds, the possibility of elimination reaction is very high.
Question 4: Which reference books can be followed to prepare for alkyl halides?
Answer: Go through the NCERT chemistry textbook part-I for class 12th, this book contains all the details in a very precise way. NCERT is the most student-friendly book as it covers all the topics that are significant from a JEE point of view. Organic Chemistry by Morrison Boyd can be used as a reference book.
Comments