The hardness of water is due to the presence of soluble bicarbonates, chlorides and sulfates of calcium and magnesium. Water which does not give lather with soap is hard water.
Download Complete Chapter Notes of Hydrogen
Download Now
Water is the most important compound that is needed for the survival of life on earth. Water is present in oceans, rivers, ponds, lakes, glaciers, etc. Rainwater is considered pure water because it does not contain any salt dissolved in it though there are dissolved gases present.
JEE Main 2021 LIVE Chemistry Paper Solutions 24-Feb Shift-1 Memory-based

Water can be classified as hard water and soft water.
- Soft water: It lathers with soap. Water which is obtained from the rains is soft water. This water is suitable for household purposes, for example, laundry and cleaning.
- Hard water: It is known as hard water because of the presence of calcium and magnesium salts. Hard water does not lather with soap but instead forms a precipitate.
⇒ Check: Differences between Hard Water and Soft Water
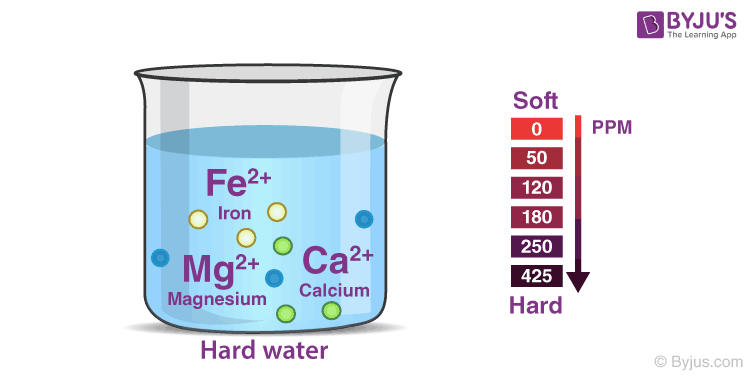
Table of Content
- What Is Hard Water?
- Types of Hardness of Water
- Disadvantages
- Remove Temporary Hardness
- Remove Permanent Hardness
- Harmful Effects of Hard Water
What Is Hard Water?
Hard water has high mineral content. It is formed when water percolates through the deposits of chalk and limestone, which are made up of magnesium and calcium carbonates. It does not lather with soap, so it is not suitable for laundry purposes.
The hardness of water is harmful to the boilers as the deposition of salts occurs, which reduces the efficiency of the boiler. Hard water is safe to drink, but using it over a long interval of time can lead to many problems like:
- Strains in skin
- Water appliances work harder, resulting in higher water bills
- Spots appear on clothes and linens
Types of Hardness of Water
Hardness of water can be classified into two types:
- Temporary Hardness
- Permanent Hardness
Temporary Hardness of Water
The presence of magnesium and calcium carbonates in water makes it temporarily hard. In this case, the hardness in water can be removed by boiling the water.
When we boil water, the soluble salts of Mg(HCO3)2 are converted to Mg(OH)2, which is insoluble, and hence gets precipitated and is removed. After filtration, the water we get is soft water.
Permanent Hardness of Water
When the soluble salts of magnesium and calcium are present in the form of chlorides and sulphides in water, we call it permanent hardness because this hardness cannot be removed by boiling.
We can remove this hardness by treating the water with washing soda. Insoluble carbonates are formed when washing soda reacts with the sulphide and chloride salts of magnesium and calcium, and thus, hard water is converted to soft water.
Disadvantages of Hardness
- Wastage of soap
- Wastage of fuel
- Formation of scales on metallic boilers.
⇒ Also Read: Heavy Water – Methods of Preparation and Properties
Remove the Hardness of Water (Temporarily)
By Boiling:
Soluble bicarbonates are converted into insoluble carbonates, which are removed by filtration.
Reactions: Ca(HCO3)2 → ΔCalo3↓ + H2O + CO2
⇒ Mg(HCO3)2 → ΔMgCO3↓ + H2O + CO2
By Clarks Method:
Calcium hydroxide is Clark’s reagent. It removes the hardness of water by converting bicarbonates into carbonate.
Reaction: Ca(OH)2 + Ca(HCO3)2 → 2CaCO3↓ + 2H2O
How to Remove Permanent Hardness of Water?
Gan’s Permutit Method:
In this method, sodium aluminium ortho silicate, known as permutit or zeolite, is used to remove the permanent hardness of the water.
Reaction: Na2 Al2 Si2 O8.KH2O + Ca++→ 2Na+ + Ca Al2 Si2 O8.xH2O
Calgon’s Process:
In this method, sodium-hexa-meta-phosphate (NaPO3)6, known as Calgon, is used. The hardness in water is removed by the adsorption of Ca++ and Mg++ ions.
⇒ Check: Difference between Adsorption and Absorption
Ion Exchange Resin Method:
In this method, the permanent hardness of water is removed by using resins. Ca++/Mg++ ions are exchanged with Cl–, and SO4-2 ions are exchanged with anion exchange resin (RNH2OH). Demineralised water is formed in this process.
⇒ 2RCOOH + Ca++ → (RCOO)2Ca + 2H+
⇒ RNH2OH + Cl– → RNH2Cl + OH–
⇒ H+ + OH– → H2O
Harmful Effects of Hard Water
Some of the most common signs of hard water include the following:
- Linens and clothes look dull and feel rough.
- Ugly stains on white porcelain and scale build-up on faucets
- Low water pressure from showers due to clogged pipes.
- Chalky, white residue or spots appear on dishes.
- Strains appear in the shower.

Comments