
If you are studying chemistry, then you must have wondered about the importance of chemistry in everyday life. Chemistry is the branch of science which deals with the investigation of the properties and changes of matter. From the way how our body exchanges oxygen to how our universe was created, all have a part of chemistry associated with it.
Download Complete Chapter Notes of Chemistry in Everyday Life
Download Now
Importance of Chemistry in Everyday Life
Read More:
Chemicals of Food in Everyday Life
The following chemicals are widely used in food materials.
- Colouring agents
- Artificial preservatives
- Flow stabilisers
- Binding substance
- Artificial sweetness
- Antioxidants
- Minerals
- Vitamins
These substances do not have nutritional value except vitamins.
Also Read: Important Questions on Chemistry in Everyday Life
Artificial Preservatives: They prevent spoilage of food by stopping the growth of microorganisms. For example, sodium benzoate and sodium meta bisulphate.
Artificial Sweetness: They do not impart any calories to the body since these substances are excreted through urine. For example,
- Aspartame: It is used in cool drinks and ice creams.
- Alitame: It is 2000 times sweeter than sucrose.
Antioxidants: They prevent the spoilage of food by preventing the oxidation of food. For example,
- Butylated hydroxyl tolerance (BHT)
- Butylated hydroxyl anisole (BHA)
Dyes are coloured organic compounds that are used to impart colour to various substrates, including paper, leather, fur, hair, drugs and cosmetics. Dyes are classified into natural dyes and synthetic dyes.
Chemistry of Cleansing Agents in Everyday Life
What are soap and detergents?
Soaps are sodium or potassium salt of higher carboxylic acid such as stearic acid, palmitic acid and oleic acid, whereas detergents contain a long chain of alkyl groups. Detergents, in comparison to soaps, can also function in hard water.
Saponification: Alkaline hydrolysis of triesters of glycerol to form soap is known as saponification. Soap does not function in hard water since they precipitate in it.
How do soaps work?
Soaps are generally sodium or potassium salts of long-chain fatty acids. Soap molecules have a hydrophobic as well as a hydrophilic part. While the hydrophilic part clings to the water when washing, the hydrophobic end clings to the dirt particles. Thus, when we pour away the water, the dirt particles wash away with the soap molecules.
Also Read: Cleansing Action of Soaps and Detergents
Types of Soaps
- Toilet Soaps: Potassium soaps are softer than sodium soaps.
- Floating Soaps: They can be prepared by beating soap bubbles.
- Transparent Soaps: They contain soap dissolved in excess of alcohol, and it is evaporated.
- Medicated Soaps: They contain soaps by adding little amounts of Dettol, Savlon, etc.
- Laundry Soaps: They mainly contain sodium rosinate and borax.
Types of Detergents
Anionic Detergent: In this type, anions act as detergents. For example, sodium lauryl sulphate
Cationic Detergent: In this type, cations act as detergents. For example, cetyl trimethyl ammonium bromide.
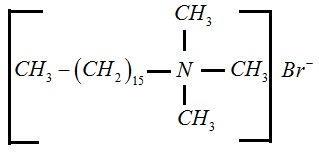
Non-ionic Detergent: They are neutral. The whole molecule acts as a detergent. For example, polyethylene glycol stearate.
Chemistry of Cosmetics in Everyday Life
Cosmetics contain the following categories of chemicals:
- Emulsifiers: They increase the stability of the emulsion. For example, potassium cetyl sulfate.
- Preservatives: They are added to cosmetics to increase their shelf life. For example, benzyl alcohol and salicylic acid.
- Thickeners: They give an appealing consistency. For example, cetyl alcohol and stearic acid.
- Emollients: They soften the skin by preventing water loss. For example, glycerine and zinc oxide.
- Glimmer and Shiners: For example, mica, bismuth oxychloride.
Other Examples of Chemistry in Everyday Life
Let us now discuss some common examples of chemistry in everyday life which most of us never knew about.
The Expiration Date on Bottled Drinking Water
Have you ever wondered why there is an expiration date on a bottle of drinking water? After all, it is just water, isn’t it? Well, most of us haven’t even noticed that there is, in fact, an expiration date on the bottle. The idea behind instilling an expiration on bottled drinking water is to standardise its packaging quality.
What the actual expiration date signifies is if the expiration date is up, the taste of the water will be different as there is a chance that the chemicals in the packaging material may ruin the quality of the water.
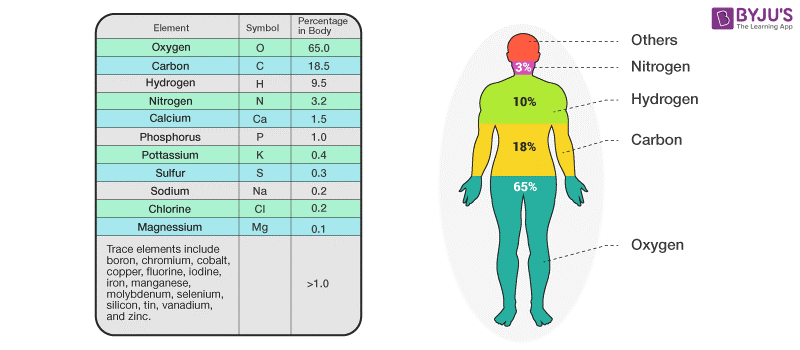
Elements in the Human Body
We all know that our body is about 60% water, but then what composes the rest of it? Carbon, Hydrogen, Nitrogen and Oxygen. These elements compose 96% of the human body. Whereas the rest 4% is composed of about 60 elements. Some of these elements include calcium, phosphorus, potassium and sulphur.
Sunblock and Sunscreen
There are two kinds of rays from the sun which are particularly bad for us, UV-A and UV-B. A sunscreen’s action, as the name suggests, functions as a screen and offers protection from sunburn which is caused by UV-B. Whereas, a sunblock has more of reflective nature and blocks both UV-A and UV-B radiations.

Comments