NCERT Solutions for Class 10 Science Chapter 4 – CBSE Free PDF Available
NCERT Solutions for Class 10 Science Chapter 4 – Carbon and Its Compounds helps students to understand concepts provided in the textbook in detail. NCERT Solutions for Class 10 Science provides solutions to all the questions asked at the end of every chapter and the questions printed within a chapter.
Download Exclusively Curated Chapter Notes for Class 10 Science Chapter – 4 Carbon and Its Compounds
Download Most Important Questions for Class 10 Science Chapter – 4 Carbon and Its Compounds
These NCERT Solutions for Class 10 Science are designed by our subject experts, who are highly experienced. The NCERT Solutions created are authentic and can be referred to by the students as a guide to prepare for the CBSE Board examination. The content is in simple language, which makes it easy to understand.
The concepts are explained with diagrams, points to remember, step-by-step procedures, shortcuts for numerical problems and tricks to remember formulae and chemical reactions. These NCERT Solutions provide an overview of the main concepts in the chapter and help you to get well-versed in important topics such as writing chemical equations and balancing them. Students can download the solutions PDF for free from the links given below.
NCERT Solutions for Class 10 Science Chapter 4 Carbon and Its Compounds
Access Answers to NCERT Class 10 Science Chapter 4 – Carbon and Its Compounds
(All In-text and Exercise Questions Solved)
In-text questions set 1, page number 61
1. What would be the electron dot structure of carbon dioxide which has the formula CO2?
Solution:
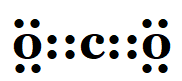
2. What would be the electron dot structure of a molecule of Sulphur which is made up of eight atoms of Sulphur? (Hint – The eight atoms of Sulphur are joined together in the form of a ring).
Solution:
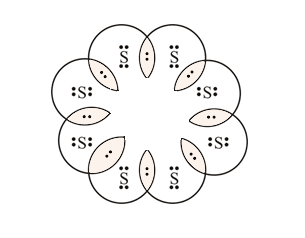
In-text questions set 2, page number 61
1. How many structural isomers can you draw for pentane?
Solution: The structural isomers of pentane are as follows:
n-pentane
2-methylbutane
2, 2-dimethylpropane
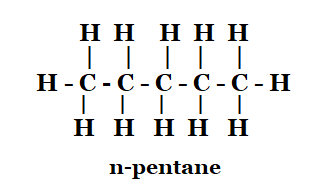
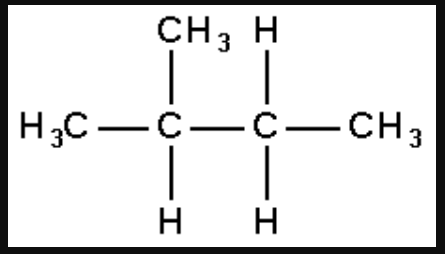
2-methylbutane
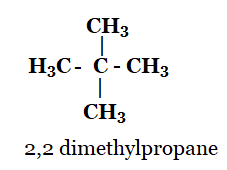
2. What are the two properties of carbon which lead to the huge number of carbon compounds we see around us?
Solution: Two properties of carbon which lead to the huge number of carbon compounds we see around us are as given below:
- Carbon has six valence electrons which is actually a high number of valency.
- Covalent bonding happens easily with carbon atoms and numerous others, such as oxygen, chlorine, nitrogen, sulphur, hydrogen, etc.
3. What will be the formula and electron dot structure of cyclopentane?
Solution: The formula and electron dot structure of cyclopentane is as given below:
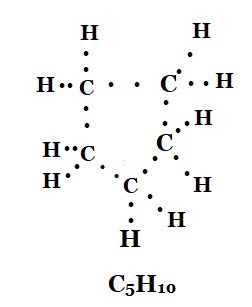
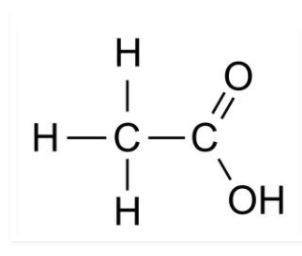
4. Draw the structures for the following compounds.
(i) Ethanoic acid
(ii) Bromopentane*
(iii) Butanone
(iv) Hexanal
Solution: i)
ii)
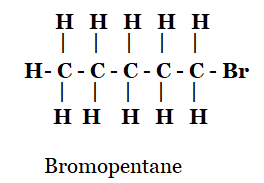
iii)
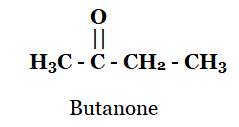
iv)
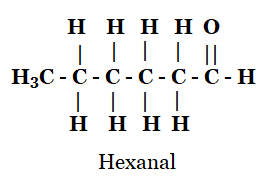
5. How would you name the following compounds?
- CH3—CH2—Br
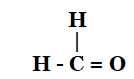
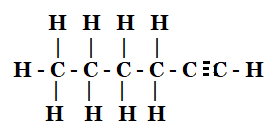
Solution:
-
- Bromoethane
- Methanal or Formaldehyde
- 1 – Hexyne
In-text questions set 3, page number 71
1. How is the conversion of ethanol to ethanoic acid an oxidation reaction?
Solution:
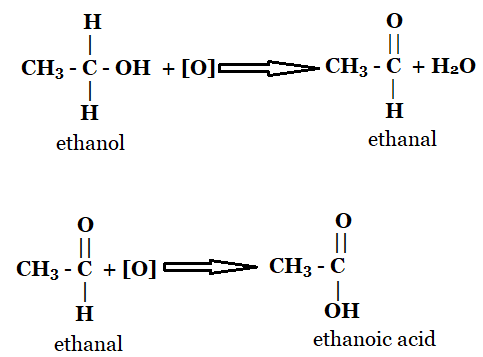
The conversion of ethanol to ethanoic acid involves the removal of the hydrogen atom and the addition of oxygen, and it is an oxidation reaction. In the first step, a H2 molecule is removed from ethanol to form ethanal. As the loss of hydrogen is oxidation, so the reaction is an oxidation reaction. Similarly, an oxygen atom is added to form ethanoic acid from ethanal. As the gain of oxygen is called oxidation, the reaction is an oxidation reaction.
2. A mixture of oxygen and ethyne is burnt for welding. Can you tell why a mixture of ethyne and air is not used?
Solution: A mixture of oxygen and ethyne is burnt for welding instead of a mixture of ethyne and air because the production of heat is very important for welding metals. When oxygen and ethyne are burnt, it burns completely and produces a higher temperature than air and ethyne. Oxygen and ethyne produce a very hot blue flame, but the mixture of air and ethyne gives out a sooty flame which means that there are unburnt particles, resulting in lesser heat.
In-text questions set 4, page number 74
1. How would you distinguish experimentally between an alcohol and a carboxylic acid?
Solution: In reaction with sodium carbonate, carboxylic acids produce carbon dioxide gas which turns lime water milky, whereas alcohols do not give this reaction. This experiment can be used to distinguish alcohol and carboxylic acid.
The reaction of the carboxylic acid with sodium carbonate:
2CH3COOH + Na2CO3 → 2CH3COONa + H2O + CO2
2. What are oxidising agents?
Solution: Oxidising agents are those compounds which either remove hydrogen or add oxygen to a compound. For example, halogens, potassium nitrate, and nitric acid.
In-text questions set 5, page number 76
1. Would you be able to check if water is hard by using a detergent?
Solution: It is not possible to check if water is hard by using a detergent because detergents are salts of ammonium or sulphonates of long-chain carboxylic acids. Unlike soaps, they do not react with calcium and magnesium to distinguish the nature of water.
2. People use a variety of methods to wash clothes. Usually, after adding the soap, they ‘beat’ the clothes on a stone, or beat it with a paddle, scrub with a brush or the mixture is agitated in a washing machine. Why is agitation necessary to get clean clothes?
Solution: Agitation is necessary to get clean clothes as it aids soap micelles to trap the oil, grease or any other impurities that have to be removed. When they are being beaten or agitated, the particles are removed from the clothes’ surfaces and go into the water, thus cleaning the clothes.
Exercise questions, page number 77-78
1. Ethane, with the molecular formula C2H6, has
(a) 6 covalent bonds
(b) 7 covalent bonds
(c) 8 covalent bonds
(d) 9 covalent bonds
Answer: (b) 7 covalent bonds
Solution: Ethane, with the molecular formula C2H6, has 7 covalent bonds.
2. Butanone is a four-carbon compound with the functional group
(a) carboxylic acid
(b) aldehyde
(c) ketone
(d) alcohol
Answer: (c) ketone
3. While cooking, if the bottom of the vessel is getting blackened on the outside, it means that
(a) the food is not cooked completely
(b) the fuel is not burning completely
(c) the fuel is wet
(d) the fuel is burning completely
Answer: (b) the fuel is not burning completely
Solution: While cooking, if the bottom of the vessel is getting blackened on the outside indicates that the fuel is not burning completely.
4. Explain the nature of the covalent bond using the bond formation in CH3Cl.
Solution: Carbon can neither lose 4 electrons nor gain four electrons as these processes make the system unstable due to the requirement of extra energy. Therefore, CH3Cl completes its octet configuration by sharing its 4 electrons with carbon atoms or with atoms of other elements. Hence, the bonding that exists in CH3Cl is a covalent bonding.
Here, carbon requires 4 electrons to complete its octet, while each hydrogen atom requires one electron to complete its duplet. Also, chlorine requires an electron to complete the octet. Therefore, all of these share electrons, and as a result, carbon forms 3 bonds with hydrogen and one with chlorine.
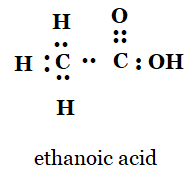
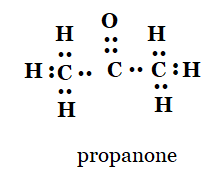
5. Draw the electron dot structures for
(a) ethanoic acid
(b) H2 S
(c) propanone
(d) F2
Solution:
a)
b)
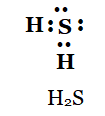
c)
d)
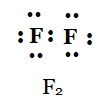
6. What is a homologous series? Explain with an example.
A homologous series is a series of compounds which has the same functional group. This also contains a similar general formula and chemical properties. Since there is a change in the physical properties, we can say that there would be an increase in molecular size and mass.
For example, methane, ethane, propane, butane, etc., are all part of the alkane homologous series. The general formula of this series is CnH2n+2. Methane CH4 Ethane CH3CH3 Propane CH3CH2CH3 Butane CH3CH2CH2CH3. It can be noticed that there is a difference of −CH2 unit between each successive compound.
7. How can ethanol and ethanoic acid be differentiated on the basis of their physical and chemical properties?
Solution:
| Ethanol | Ethanoic acid |
| It does not react with sodium hydrogen carbonate | Bubbles and fizzes with sodium hydrogen carbonate |
| A good smell | Smells like vinegar |
| No action in litmus paper | Blue litmus paper to red |
| Burning taste | Sour taste |
8. Why does micelle formation take place when soap is added to water? Will a micelle be formed in other solvents, such as ethanol also?
Solution: Micelle formation takes place because of the dirt particles in water and clean water. There are two mediums that are involved: one is pure water, and the other is dirt, also called impurities. The soap also has two mediums:
(i) organic tail
(ii) ionic head
So the organic tail mixes and dissolves with the dirt, whereas the oil or grease and ionic head dissolve and mix with the water. Therefore, when the material to be cleaned is removed from the water, the dirt is taken off by the soap molecules in the water. Hence, the soap cleans by forming closed structures through the mutual repulsion of the micelles (positively charged heads).
Other solvents, such as ethanol, in which sodium salt of fatty acids does not dissolve, so not able to form such micelles.
9. Why are carbon and its compounds used as fuels for most applications?
Solution: Carbon and its compounds are used as fuels for most applications because they have high calorific values and give out a lot of energy. Most of carbon compounds give a lot of heat and light when burnt in the air.
10. Explain the formation of scum when hard water is treated with soap.
Solution: Scum is produced from the reaction of hard water with soap. Calcium and magnesium present in the hard water form an insoluble precipitate called scum.
11. What change will you observe if you test soap with litmus paper (red and blue)?
Solution: When soap is dissolved in water due to the formation of alkaline NaOH or KOH, the solution is alkaline. The solution changes the colour of the red litmus to blue, but in the soap solution, the blue litmus remains blue.
12. What is hydrogenation? What is its industrial application?
Solution: Hydrogenation is a process or a chemical reaction between hydrogen and other compounds. It is usually done in the presence of catalysts. For example, nickel, palladium or platinum. Hydrogenation is used mainly to saturate organic compounds.
13. Which of the following hydrocarbons undergo addition reactions: C2H6, C3H8, C3H6, C2H2 and CH4?
Solution: Unsaturated hydrocarbons undergo addition reactions. C3H6 and C2H2 are unsaturated hydrocarbons which undergo addition reactions.
14. Give a test that can be used to differentiate between saturated and unsaturated hydrocarbons.
Solution: The bromine water test is used to differentiate between the unsaturated compounds (like alkenes and alkynes) and the saturated compounds. For this purpose, bromine is used in the form of bromine water. A solution of bromine in water is called bromine water. Bromine water has a red-brown colour due to the presence of bromine in it. When bromine water is added to an unsaturated compound, then bromine gets added to the unsaturated compound, and the red-brown colour of bromine water is discharged. So, if an organic compound decolourises bromine water, then it will be an unsaturated hydrocarbon (containing a double bond or a triple bond), but saturated hydrocarbons (alkanes) do not decolourise bromine water.
The bromine water test is performed to differentiate between the unsaturated compounds (like alkenes and alkynes) and the saturated compounds. When bromine water is added to an unsaturated hydrocarbon, the red-brown colour of the bromine solution is discharged. So, if there is dis-colouration, then the compound will be an unsaturated hydrocarbon.
15. Explain the mechanism of the cleaning action of soaps.
Solution: There are so many impurities and dirt mixed in water, and most of all, the dirt does not dissolve in the water. Soap molecules are a combination of salts such as sodium or potassium. The molecules are of a long chain of carboxylic acids. So, when the carbon chain is dissolved in oil, and the ionic end is dissolved in the water, the soap starts cleansing and trapping the dirt. When this happens, the soap molecules form structures called micelles that are used for capturing the oil droplets, and then the other end is the ionic faces. This will then form an emulsion in water and help in dissolving the dirt or impurities when the clothes are washed.
The soap molecules have different properties at different ends. The first end is the hydrophilic end, which dissolves in the water and is attracted towards the water, and the second one is the hydrophobic end, which is dissolved in the hydrocarbons and is repulsive to water. The hydrophobic tail aligns itself along the surface of the water because it is not soluble in the water.
| Also Access |
| NCERT Exemplar Solutions for Class 10 Science Chapter 4 |
| CBSE Notes for Class 10 Science Chapter 4 |
NCERT Solutions for Class 10 Science Chapter 4 – Carbon and Its Compounds
The average number of questions usually asked from this chapter is 2+3 marks. In the year 2017, a total of 15 mark questions were asked from this chapter, whereas in the year 2018, the weightage was 2+3 = 5.
List of Section Questions with Type
| Section Number | Section Name | Questions | Question Type |
| Section 4.1 | Bonding in carbon – The covalent bond | 2 | 2 short answers |
| Section 4.2 | Versatile nature of carbon | 5 | 2 very short answers, 3 long answers |
| Section 4.3 | Chemical properties of carbon compounds | 2 | 2 long answers |
| Section 4.4 | Some important carbon compounds – ethanol and ethanoic acid | 2 | 1 short answer, 1 long answer |
| Section 4.5 | Soaps and detergents | 2 | 1 very short answer, 1 long answer |
List of Exercise Questions with Type
This chapter consists of 15 questions and is divided into the following types –
| Multiple-choice questions (MCQ) | 3 |
| Short answer (SA) | 2 |
| Long answer (LA) | 7 |
| Very long answer (VLA) | 3 |
NCERT Solutions for Class 10 Science Chapter 4 – Carbon and Its Compounds
Carbon is the basis for all living organisms and a versatile element. It is tetravalent and has the property of catenation. Carbon forms covalent bonds by sharing electrons between two atoms and achieves completely filled outermost shell. It forms covalent bonds with oxygen, chlorine, hydrogen, nitrogen, sulphur and itself. It can form double and triple-bond compounds. There exist three types of carbon chains viz, branched, ring and straight. Carbon is considered as a major source of fuel. Ethanoic acid and ethanol are carbon compounds which are important and used in our daily lives. The behaviour of detergents and soaps is because of the hydrophilic and hydrophobic groups, which help in the emulsification of oily dirt and remove it.
Key Features of NCERT Solutions for Class 10 Science Chapter 4 – Carbon and Its Compounds
- The information given in these NCERT Solutions for Class 10 Science Chapter 4 – Carbon and Its Compounds is authentic and simple.
- These solutions provide answers to all the exercise questions at the end of Chapter 4, Carbon and Its Compounds, from NCERT Class 10 Science textbook.
- The solutions to questions printed between the lesson have also been provided.
- NCERT Solutions for Class 10 Science Chapter 4 – Carbon and Its Compounds are provided by expert teachers after expansive research.
- These solutions will be useful to prepare for the CBSE exam as well as various competitive exams.
- Students can refer to these solutions to prepare for their CBSE Board exam as it consists of step-by-step procedures, neatly labelled diagrams, shortcuts, and tips to tackle complex type of questions smartly.
You can check out NCERT Solutions for Class 10 to get NCERT Class 10 chapter-wise solutions for other subjects.
Frequently Asked Questions on NCERT Solutions for Class 10 Science Chapter 4
What types of questions are present in Chapter 4 of NCERT Solutions for Class 10 Science?
1. Multiple-choice questions – 3 questions
2. Short answer – 2 questions
3. Long answer – 7 questions
4. Very long answer – 3 questions
List out the topics in Chapter 4 of NCERT Solutions for Class 10 Science.
1. Bonding in carbon – The covalent bond
2. Versatile nature of carbon
3. Chemical properties of carbon compounds
4. Some important carbon compounds – ethanol and ethanoic acid
5. Soaps and detergents













Comments